Research Projects
Dr. Somsak Ruchirawat
Chulabhorn Research Institute

Current target areas include anti-malarial, chemopreventive and anti-cancer compounds, isoquinolines and isoquinolines-derived compounds. A student might be involved in the synthesis of analogs, using the already developed synthetic route, for subsequent in-house biological screening. Students might also be involved in developing synthetic methodology to form various heterocycles. Other on-going projects include using hypervalent iodine for various oxidative transformations.
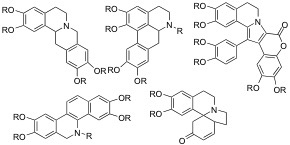
In natural products chemistry, the emphasis is on the extraction of active natural products from indigenous Thai plants as well as from fungi and marine sources. The work involves extraction, isolation, and structural elucidation employing various spectroscopic techniques, and biological evaluation as well as structure modification of the natural products. Specific examples of project include: synthesis of benzazepine alkaloids, or atom economic synthesis of protoberberine alkaloids.
Dr. Poonsakdi Ploypradith
Chulabhorn Research Institute
The Ploypradith lab has developed synthetic routes to the lamellarins, a class of bioactive compounds with anticancer and multi-drug resistance reversal activities. The ThaiREU students can work on synthesis to explore structure-activity relationships as well as the more detailed mechanisms of action in anticancer and multidrug-resistance reversal and anti-HIV-1 integrase action. Improving physic-chemical properties

(ex. aqueous solubility) of new analogs is the main focus.
Lewis/Brønsted acid catalyzed generation and trapping of quinone methide intermediates towards the total synthesis of compounds having a 2-arylchroman
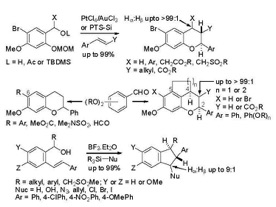
core are actively being pursued. Also, cyclization of stilbenylmethanols using Lewis/ Brønsted acids provide indanyl cations; subsequent nucleophilic transfer from the trialkylsilyl reagents provide substituted indanes. Examples of projects for the ThaiREU students would be to investigate the substrate scopes and the reaction mechanisms. In addition, total syntheses of compounds with structures containing these cores are actively pursued in our lab and the students can take part in one of these total synthesis efforts.
Dr. Charnsak Thongsornkleeb
Chulabhorn Research Institute
Dr. Pitak Chuawong
Kasetsart University
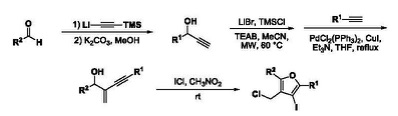
We have developed simple protocols for conducting regioselective hydrobromination of terminal alkynes and metal-free electrophilic aromatic halogenation using TMSCl and N¬-halosuccinimide. Students may be involved in developing the intramolecular cyclization of o-alkynylanilines to form both indoles and quinolines.
Dr. Thongsornkleeb investigates the cyclization processes of functionalized substrates to develop efficient methodologies towards heterocyclic scaffolds widely found in natural compounds with significant biological interest. A general strategy involves intramolecular cyclization of enynyl alcohols onto the tethered alkyne activated by electrophilic ICl.

The Chuawong lab’s research focus is on non-discriminating aspartyl-tRNA synthetase, a crucial enzyme found in organisms lacking asparaginyl-tRNA synthetase. Two ongoing projects include: domain splitting and Native Chemical Ligation in the ND-AspRS from the human pathogen Helicobacter pylori: each domain
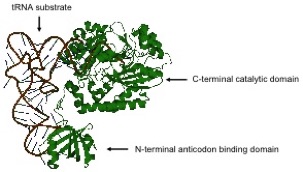
is independently overexpressed and purified to homogeneity by affinity chromatography. The resulting proteins are subjected circular dichroism, dynamic light scattering, and NMR spectroscopy to gain structural information. The ThaiREU student could work on tRNA binding studies of the ND-AspRS from Helicobacter pylori, or synthesis of substrate analogs for Asp-tRNAAsn/Glu-tRNAGln amidotransferase (GatCAB).
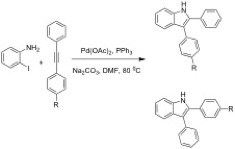
The second project involves synthesis of indoles by Larock heteroannnulations. The synthesis of 2,3-disubstituted indole compounds utilizing catalytic Pd forms a C-C bond and a C-N bond in a single reaction. We have developed 2,3-diarylindoles with subtituents in the para position, which exhibit anticancer activity against human lung cancer cells (A549). The ThaiREU student could work on the synthesis of new indole derivatives to enhance anti-cancer activity.
Dr. Nopporn Thasana
Chulabhorn Research Institute
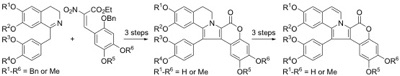
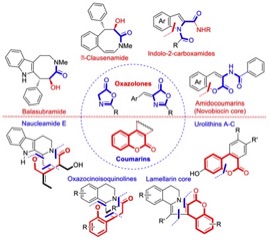
Research in the Thasana lab focuses on the synthesis of natural bioactive lactones and lactams using Cu(I)-mediated, MW-assisted C-O/C-N bond formation under “subcritical water” (green) conditions to synthesize benzopyranones (urolithins A-C),
novobiocin, and indolo-2-carboxamide analogs. The chemistry of oxazolone and coumarin derivatives has been studied to develop efficient synthetic routes for a wide range of structurally complex natural products using metal catalysis or metal-free condition in the key step. The natural bioactive alkaloids lamellarin, clausenamide, and naucleamide derivatives are current targets.
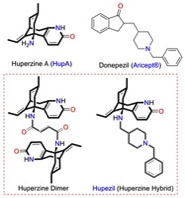
Another project focuses on screening and isolation of Lycopodium alkaloids, particularly huperzine A (HupA), from club mosses, belonging to Lycopodiaceae and Huperziaceae families. These show acetylcholinesterase (AChE) inhibition in biological screens. A summer ThaiREU student may be involved in the synthesis of Lycopodium alkaloid derivatives following methodology developed in this lab, or in the synthesis of huperzine dimer and huperzine hybrid to improve of the biological activity.

Dr. Thawatchai Tuntulani
Chulalongkorn University

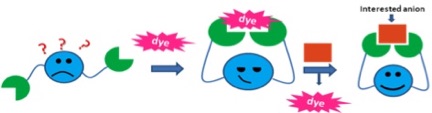
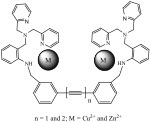
Dr. Tuntulani’s research is focused on indicator displacement assays to provide simple, convenient “naked-eye” anion sensors. An indicator is bound to a receptor by non-covalent interactions. Replacement by the analyte anion causes a change in spectroscopic signal. In these sensors, the designed receptor contains a tripodal amine linked to a diaryl mono- or diacetylene unit as a spacer. The tripodal amine can bind metal ions such as Zn2+ and Cu2+ cations, which can coordinate to the dye or the anion of interest.
The resulting competition between the indicator and the anion guest for binding is monitored by a change in the indicator’s spectral properties. The binding can be controlled by selection of the metal ion and the spacer. Students will prepare novel sensors using the general synthetic routes developed in this lab, characterize their products by 1H-NMR, 13C-NMR, elemental analysis and X-ray crystallography, and then investigate the sensing abilities of their compounds.
Dr. Tirayut Vilaivan
Chulalongkorn University
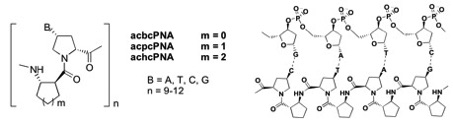
The Vilaivan lab works on "Peptide Nucleic Acids" (PNA): a DNA mimic with a peptide-like backbone that can specifically recognize nucleic acid targets (DNA, RNA) with high affinity and sequence specificity, while being completely resistant to nucleases. The focus is on α/β-peptides derived from proline (“pyrrolidinyl PNA”). Work is directed towards a better understanding of the structural aspects of the binding, and designing new PNA systems with enhanced binding properties, as well
as exploring potential applications in diagnostics and therapeutics. Examples of projects include 1) synthesis and evaluation of DNA/RNA binding properties of new pyrrolidinyl PNA systems with modified backbones/nucleobases; 2) synthesis of functional PNA probes that can cleave DNA/RNA or form covalent adducts 3) synthesis and fluorescence studies of dye-labeled acpcPNA to make fluorescent PNA probes responsive to the presence or absence of the correct DNA target. Students will gain experiences in both traditional synthesis for making PNA monomers and solid phase synthesis for making PNA oligomers. They will purify/characterize PNA by HPLC / MALDI-TOF MS, and study PNA-DNA interactions by UV-Vis, fluorescence and circular dichroism.

Dr. Chutima Kuhakarn
Mahidol University
The Kuhakarn group is focused on the development of new synthetic applications of molecular iodine and hypervalent iodine reagents, which are environmentally benign substitutes for metallic-based oxidizing reagents. They exhibit selectivity, operate under mild reaction conditions with ease of handling, and are commercial availability or readily prepared from commercial reagents. Recent published work includes the development of the oxidative functionalization of alkenes and functionalization of heteroaromatic compounds. Other research areas include organofluorine chemistry,
natural product chemistry, and the development of new synthetic methodologies. At present, reactions of difluoro-phenylsulfanylmethane mediated by Lewis acids, decarboxylative functionalization reactions, functionalization of indole derivatives, synthesis of β-ketosulfones and α-arylsulfanylketones are in progress. ThaiREU students would potentially work in any of the projects currently being carried
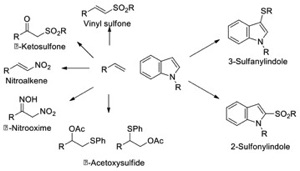

out in our laboratory. Students are expected to learn laboratory planning, reaction monitoring, separation techniques and spectroscopic analysis of the resulting products.

Dr. Tienthong Thongpanchang
Mahidol University
Dr. Thongpanchang’s lab works in two main areas. Students might be involved in either of these.
2) The design and synthesis of novel sulfur containing organic materials for organic light emitting diodes and dye sensitized solar cell applications. For example, a series of isomeric naphthodithiopenes have been prepared in two
1) The design and synthesis of new chiral derivatizing agents for determination of the absolute configuration of chiral compounds, such as THENA based on Mosher’s acid with a constrained anisotropic group, leading to an unambiguous chemical shift difference (Δδ) in NMR. The synthesis utilizes a Diels–Alder
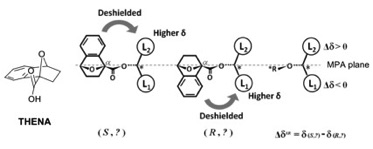
reaction and benzyne to generate a bicyclic structure. Both enantiomers can react with a chiral alcohol to form a pair of diastereomers. A ThaiREU project would involve the synthesis of THENA derivatives and application to the determination of absolute configuration.
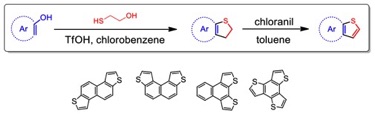
straightforward steps. A ThaiREU student would utilize new thiophene derivatives to prepare naphthodithiopenes: the optical and electronic properties would investigated by UV spectroscopy and cyclic voltammetry.
Dr. Supa Hannongbua
Kasetsart University
Dr. Hannongbua’s laboratory is interested in drug discovery for anti-AIDS treatments and tropical diseases. A computational drug discovery project includes structural modification of natural products and organic syntheses, QSAR, quantum chemical calculations, virtual screening, de novo design, combinatorial library design, protein-ligand interaction simulations, large-scale Molecular Dynamics simulations, drug-likeness analysis and ADME/T (Adsorption, Distribution, Metabolism, Excretion and Toxicology) predictions. Recently, a next generation anti-AIDS non-nucleoside reverse transcriptase inhibitor has been successfully designed in the Hannongbua lab. The ThaiREU student will be involved in the synthesis of lead compounds as well as understanding enzyme-inhibitor interactions, based on computer-aided drug design and molecular modeling techniques such as molecular docking and molecular dynamics simulations.
